The Application Gallery features COMSOL Multiphysics® tutorial and demo app files pertinent to the electrical, structural, acoustics, fluid, heat, and chemical disciplines. You can use these examples as a starting point for your own simulation work by downloading the tutorial model or demo app file and its accompanying instructions.
Search for tutorials and apps relevant to your area of expertise via the Quick Search feature. Note that many of the examples featured here can also be accessed via the Application Libraries that are built into the COMSOL Multiphysics® software and available from the File menu.
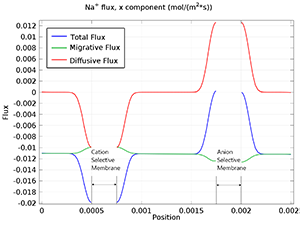
Desalination in an Electrodialysis Cell
Electrodialysis is a separation process for electrolytes based on the use of electric fields and ion selective membranes. Some common applications of the electrodialysis process are: Desalination of process streams, effluents, and drinking water pH regulation in order to remove acids ... Read More
Ion-Exchange Membranes and Donnan Potentials
This model file was used for creating the plots featured in the blog post "How to Model Ion-Exchange Membranes and Donnan Potentials". Read More
Adsorption-Desorption Voltammetry
For an electrochemical reaction to occur, the reacting species usually needs to adsorb to the electrode surface before undergoing reduction or oxidation, after which the resulting product species desorbs back into the electrolyte. If the rate of adsorption or desorption is slow in ... Read More
Potential Profile in Batteries and Electrochemical Cells
The purpose of this model is to visualize the electric potential in an electrochemical cell, for example a battery. This is done at OCV and during operation. In a battery, this would correspond to OCV, discharge, and recharge. The potential profile is explained both for cells with planar ... Read More
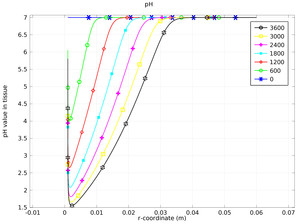
Electrochemical Treatment of Tumors
This model incorporates the transport and electrolytic reaction in the treatment of tumor tissue. Oxygen evolution at the anode produces protons, which lowers the pH, while chlorine production also leads to lowered pH through the hydrolysis of chlorine. One effect of a low pH is the ... Read More
Thin Layer Chronoamperometry
The common electroanalytical method of exhaustive amperometric detection in a microscopic thin layer is modelled as a 1D-symmetric diffusion problem. The simulated result agrees with the analytical Cottrell equation at short times, and deviates as expected at long times when the ... Read More
Isoelectric Separation
This example applies the Electrophoretic Transport and Laminar Flow interfaces to model isoelectric separation in a free-flow electrophoresis device. A stream containing six different ionic species is shown to be divided into pure component streams by means of migrative transport in an ... Read More
Zone Electrophoresis
Zone electrophoresis (ZE) is an electrophoretic separation technique typically used for analyzing proteins, nucleic acids, and biopolymers. During the process, different species in a sample are transported in a continuous electrolyte buffer system, subject to a potential gradient. Due to ... Read More
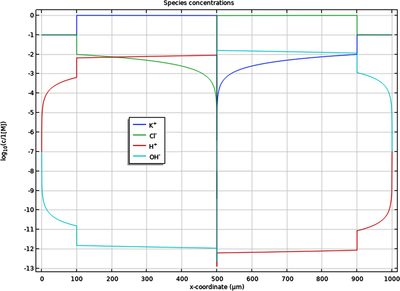
Bipolar Ion-Exchange Membrane
A bipolar membrane consists of one anion-selective, and one cation-selective membrane, in contact with each other. The combined cation and anion selectivity makes the bipolar membrane highly impermeable to all ions, with the exception of H+ and OH- which are formed by water splitting ... Read More
Lemon Battery
This asset contains the model file described in the blog post titled "Approaching an Electrochemical Model from Scratch: Lemon Battery". Read More