Modeling an Adsorption Process in a Shell-and-Tube-Heat-Exchanger-Type Adsorber
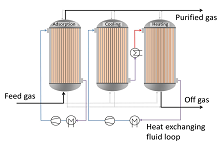
Pressure Swing Adsorption (PSA) and Temperature Swing Adsorption (TSA) are commonly used for separation/purification of gas mixtures in industrial processes. The cycle time of industrial TSA processes usually ranges from several hours to days. The reason for this long cycle time is the usage of purge gas for heating and cooling the system (direct heating), which limits the application of TSA processes to the removal of trace components. In order to reduce cycle time and to separate larger quantities of impurities a more efficient heating strategy has to be considered and a system to remove heat during adsorption has to be implemented. This can be achieved, for example, by using a shell-and-tube-heat-exchanger-type-adsorber. The cycle time can be reduced significantly by the indirect heating and cooling. Also the adsorbed component can be recovered with high concentrations since the bed is heated indirectly and not with a regeneration gas. During adsorption the released heat can be removed by a heat transfer fluid, which flows through the shell side.
The advantages obtained through the indirect heat exchange can be used for the separation of CO2 from flue gas where a relatively large amount of CO2 has to be separated. This separation process is very important to mitigate emissions of CO2 from the combustion of fossil fuels which are one of the main causes of global warming(1). Adsorption cycles using a shell and tube heat exchanger type adsorber, in which heat integration between the different vessels (hot adsorber and cold adsorber) is considered, are simulated in COMSOL Multiphysics® software. This heat integration is necessary in order to make this process energetically competitive with an amine wash as a reference process. It is the goal of this work to investigate the feasibility of this process as an alternative for carbon capture. The preliminary results are encouraging making this process an alternative for carbon capture. Nevertheless there are still some parameters that require further optimization.
Download
- duarte_poster.pdf - 0.93MB
- duarte_abstract.pdf - 0.02MB



