Simulation of Adsorption Mechanisms of Methane and Carbon Dioxide in Shale Matrix
The aim of this research is to contribute with the development of Carbon Capture and Storage techniques by studying carbon dioxide (CO2) adsorption mechanisms in shale reservoirs. Gas desorption is considered a major gas production mechanism and has a relevant role in shale gas reservoirs. In addition to methane adsorption/desorption mechanisms, there is high interest in studying CO2 adsorption to estimate the potential for Enhance Oil Recovery (EOR) as well as carbon sequestration.
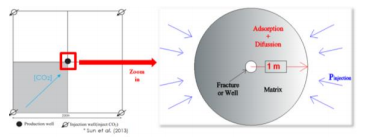
According to Kang et al. (2011), CO2 shows higher adsorption capacity than CH4. Their laboratory results successfully fit Langmuir isotherm model. Sun et al. (2013) and Prajapati and Mills (2014) modeled the flux of gas in shale matrix and coupled it with the Langmuir adsorption mechanism. Jin and Firoozabadi (2014) performed numerical simulation to explain the dynamics at molecular level during CH4 and CO2 adsorption in clay. They identified that, under water presence in the porous media, water competes with CO2 or CH4 for surface area in small nano-pores, and in other cases CO2 or CH4 form a weak second adsorption layer. This phenomenon cannot be modeled by a Langmuir adsorption isotherm. Feng et al. (2018) have measured the N2 adsorption capacity of clay samples under different water content and fit their results under a multilayer adsorption model. They inferred that the adsorption contribution to gas storage might be diminished under high water presence.
This study extends the previous works and focus in the modeling of CH4 and CO2 adsorption in shale reservoirs at micro to nano-scale by the use of COMSOL Multiphysics. The modeling of both physics, gas flux and adsorption, have been accomplished applying the Transport of Diluted Species in Porous Media module. Adsorption of CH4 and CO2 in shale matrix assuming no water content was analyzed by using Langmuir, Freundlich, and Brunauer-Emmett-Tellet (BET) Isotherm models. Freundlich and BET adsorption models are not restricted to monolayer formation and could be considered as a potential approach for modeling under water presence. These adsorption models were coupled with an equation for gas flux from the fracture to the shale particle surface. As a first approximation, the Wilke model was used to compute gas flux due to its simplicity and minimum computational requirements. Because former studies state that Knudsen diffusion should be considered for a more accurate representation of gas flux through the nano-porous matrix, Wilke-Bosanquet equation was also implemented as a second approximation for the flux modeling. Our results indicate that Langmuir and Freundlich adsorption models provide similar values. A small deviation was found at low pressures. Therefore, it was concluded that Freundlich model can be used for modeling gas adsorption in shales under conditions where the existence of monolayer is not guaranteed.
Download
- guillamon_presentation.pdf - 1.67MB
- guillamon_paper.pdf - 0.65MB
- guillamon_abstract.pdf - 0.13MB



