Galvanic Corrosion of a Magnesium Alloy in Contact with Steel
Application ID: 10914
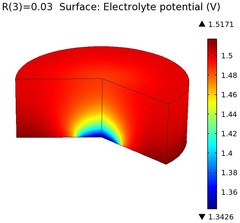
Magnesium alloys are attractive alternatives as lightweight materials in various fields of engineering. Magnesium is however relatively non-noble and may suffer considerable galvanic corrosion when being used in conjunction with other metals, for instance when mounting an Mg alloy component using steel fasteners.
This model example simulates a galvanic corrosion couple consisting of a magnesium alloy (AZ91D) and mild steel, with salt water (5% NaCl) as electrolyte. The example is based on a paper by J.X. Jia and others.
This model example illustrates applications of this type that would nominally be built using the following products:
however, additional products may be required to completely define and model it. Furthermore, this example may also be defined and modeled using components from the following product combinations:
- COMSOL Multiphysics® and
- either the Battery Design Module, Corrosion Module, Electrochemistry Module, Electrodeposition Module, or Fuel Cell & Electrolyzer Module
The combination of COMSOL® products required to model your application depends on several factors and may include boundary conditions, material properties, physics interfaces, and part libraries. Particular functionality may be common to several products. To determine the right combination of products for your modeling needs, review the Specification Chart and make use of a free evaluation license. The COMSOL Sales and Support teams are available for answering any questions you may have regarding this.



