Corrosion Processes
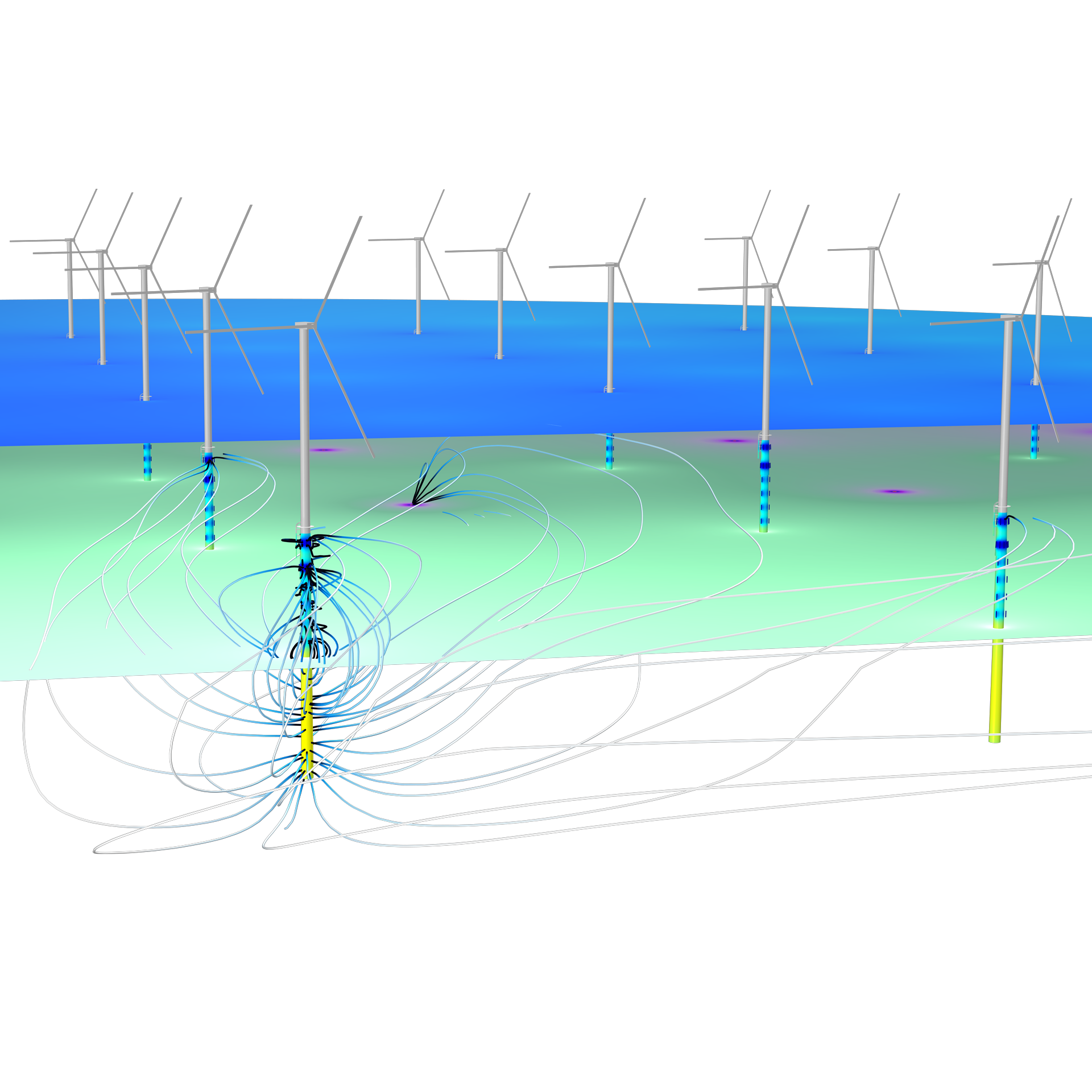
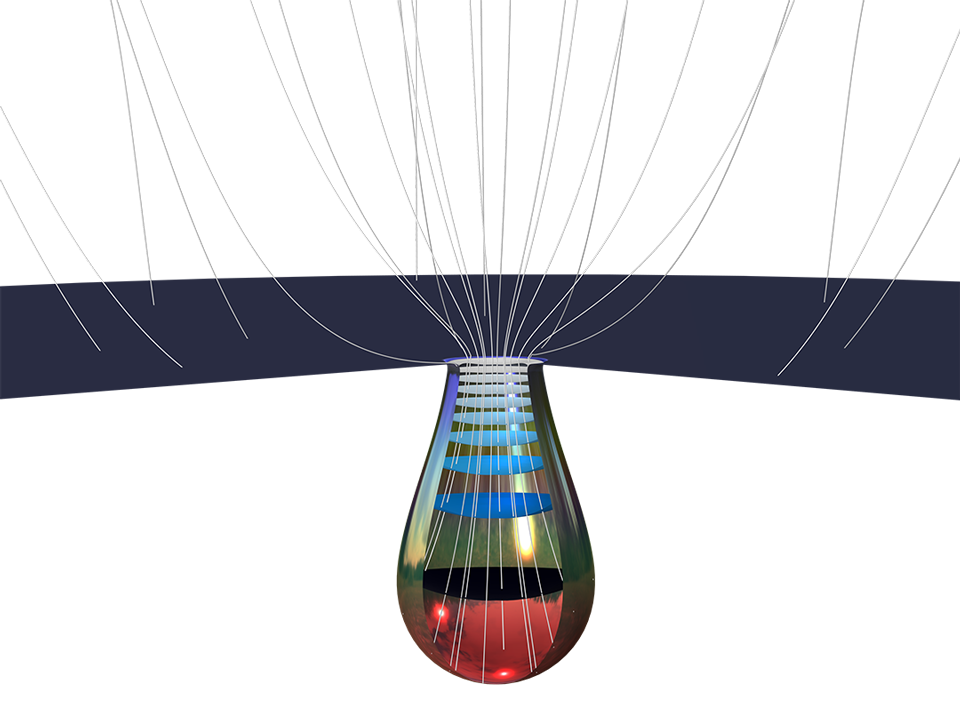
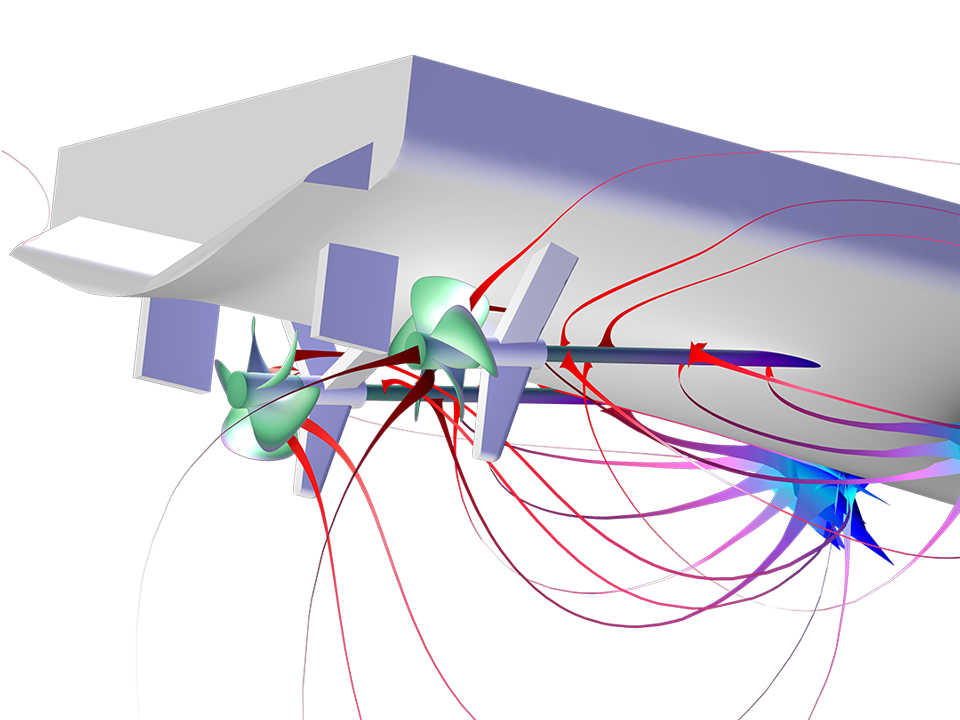
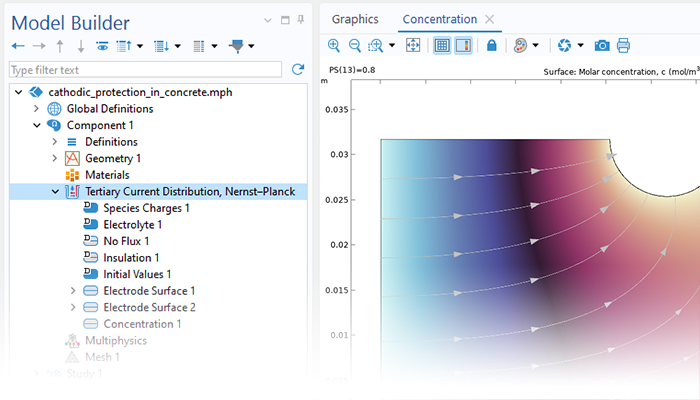
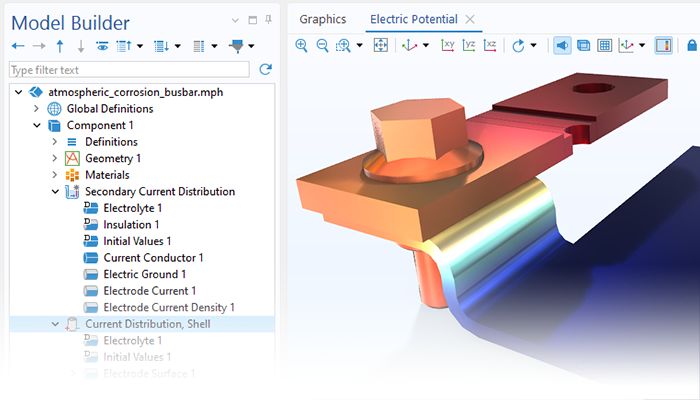
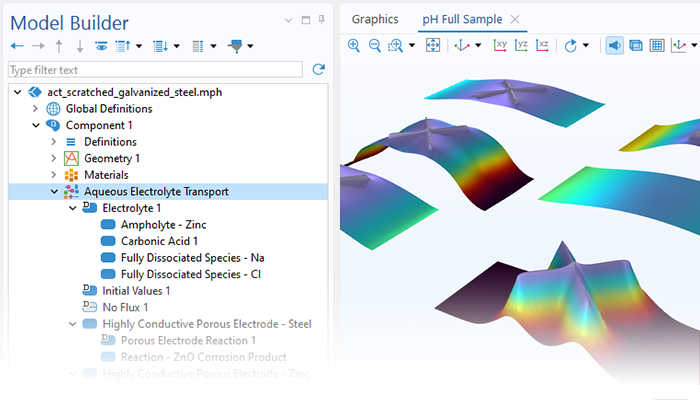
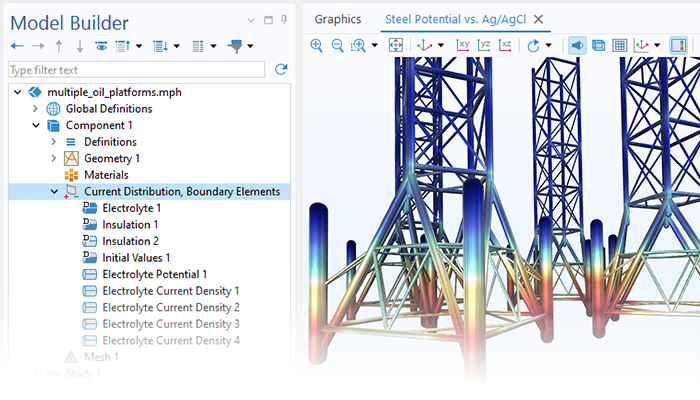
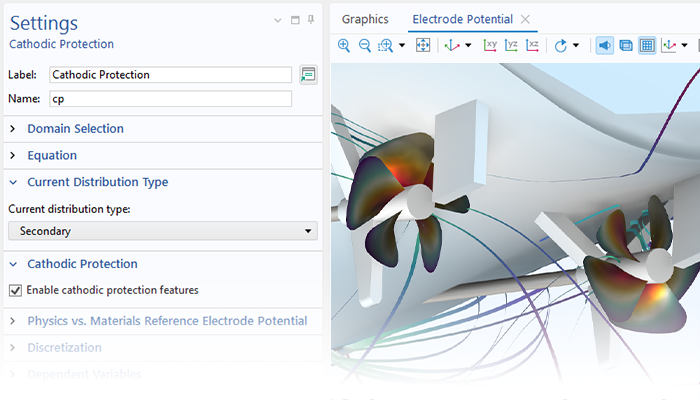
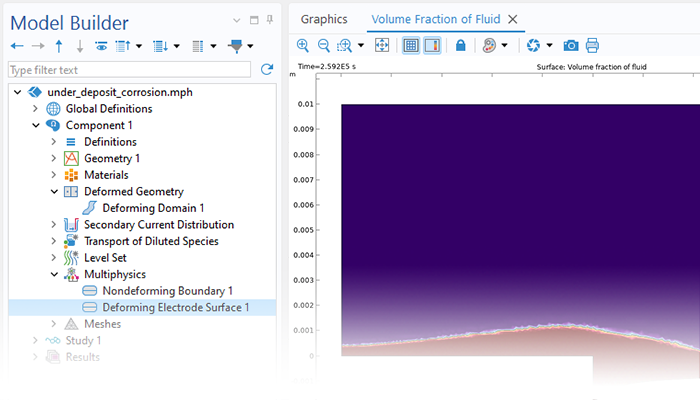
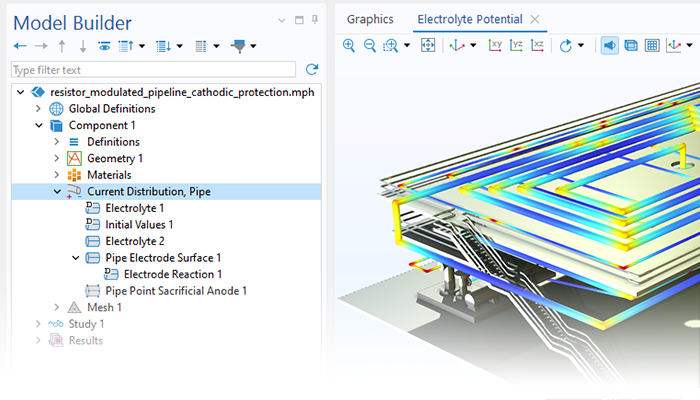
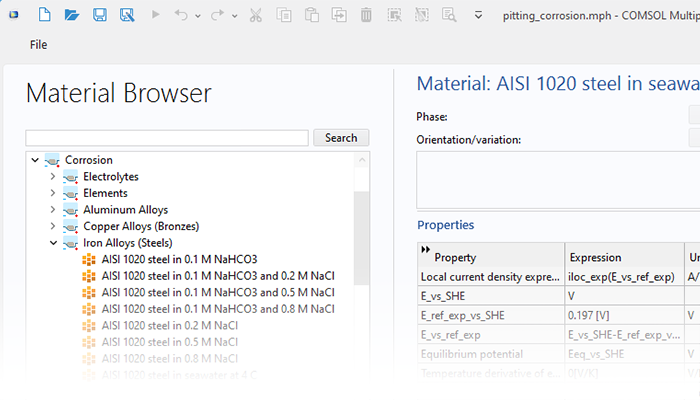
The Corrosion Module can be used to model a wide variety of corrosion processes, such as galvanic, pitting, crevice, atmospheric, and general corrosion. It can also be used to model sweet and sour corrosion in oil and gas systems, corrosion in reinforced concrete, and corrosion from stray currents.
These corrosion processes stem from similar types of electrochemical phenomena for which the transport and balance of both charge and mass must be considered. The module makes it easy to define relevant boundary conditions, surface reactions, and bulk electrolyte conditions. The electrolyte can be described as a thin layer of moisture, a porous material, or a liquid electrolyte.
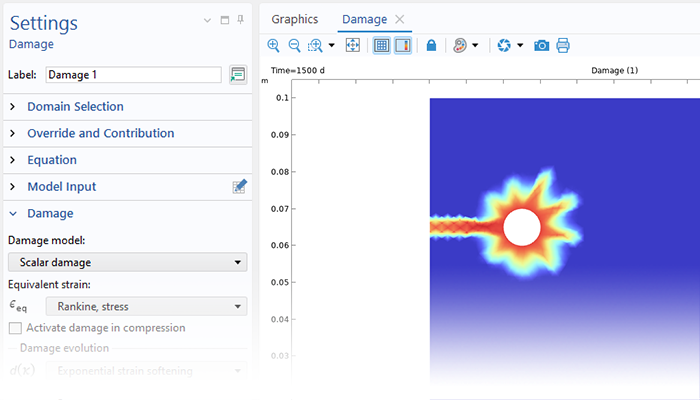
With the underlying capabilities of the COMSOL Multiphysics® platform, the Corrosion Module can be combined with other add-on products to model coupled phenomena, such as heat transfer and structural mechanics. This multiphysics modeling functionality is available for phenomena involved in problems like stress corrosion cracking (SCC) and oxide jacking in concrete.