Bipolar Ion-Exchange Membrane
Application ID: 85901
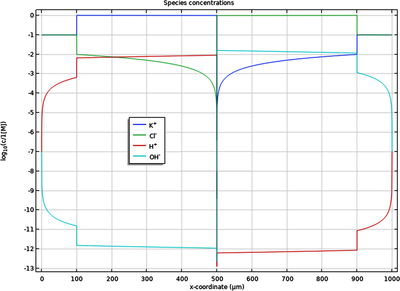
A bipolar membrane consists of one anion-selective, and one cation-selective membrane, in contact with each other.
The combined cation and anion selectivity makes the bipolar membrane highly impermeable to all ions, with the exception of H+ and OH- which are formed by water splitting (autoprotolysis) in the boundary region between the two membranes.
The dual anion/cation selectivity makes the bipolar membrane highly interesting for, for instance, water purification applications.
This model example shows how to model a bipolar membrane, including water splitting, by the use of the coupled Nernst-Planck-Poisson (NPP) equations.
This model example illustrates applications of this type that would nominally be built using the following products:
however, additional products may be required to completely define and model it. Furthermore, this example may also be defined and modeled using components from the following product combinations:
The combination of COMSOL® products required to model your application depends on several factors and may include boundary conditions, material properties, physics interfaces, and part libraries. Particular functionality may be common to several products. To determine the right combination of products for your modeling needs, review the Specification Chart and make use of a free evaluation license. The COMSOL Sales and Support teams are available for answering any questions you may have regarding this.



