Blog Posts Tagged Electrochemistry Module
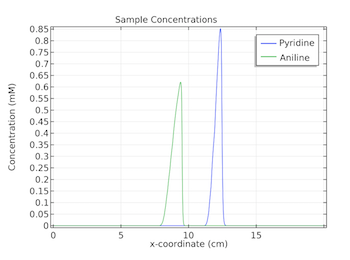
Studying Zone Electrophoresis with COMSOL Multiphysics®
Zone electrophoresis enables scientists to study nucleic acids, biopolymers, and proteins in a wide range of areas. COMSOL Multiphysics® can be used to take a closer look at this process.
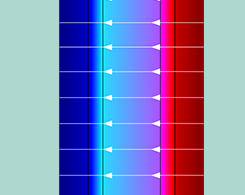
Analyzing Reverse Electrodialysis Units with Multiphysics Modeling
Pass the salt…for a clean energy solution. Salinity gradient power relies on osmosis between fresh- and saltwater to generate power, and simulation can help analyze and optimize such systems.
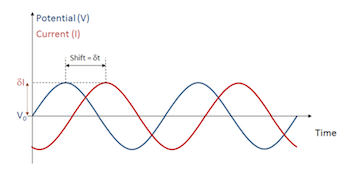
Electrochemical Impedance Spectroscopy: Experiment, Model, and App
Experience the phenomenon of electrochemical impedance spectroscopy (EIS) in 3 ways: experiment, model, and simulation application.
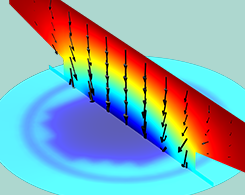
How to Model Electrochemical Resistance and Capacitance
Get an overview of the theory and modeling considerations for resistive and capacitive effects, which are fundamental to the understanding of electrochemical systems.
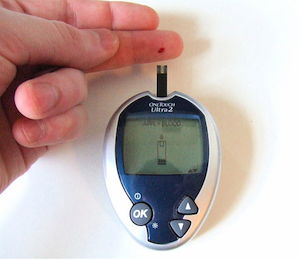
Modeling Electrochemistry for Managing Diabetes
An estimated 350 million people worldwide are living with diabetes. By modeling electrochemical processes, biomedical engineers can analyze and optimize the glucose sensing process.
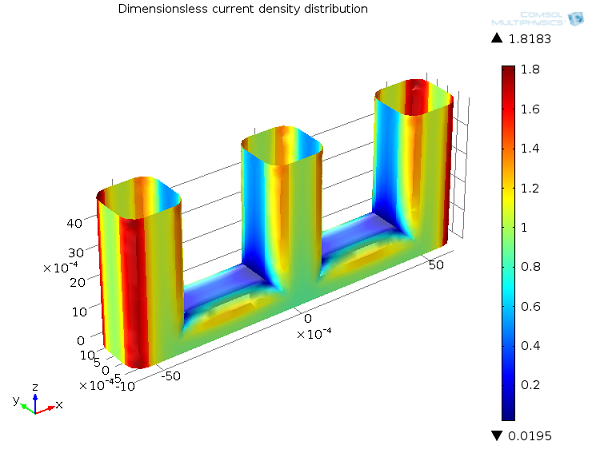
Which Current Distribution Interface Do I Use?
See the different current distributions with a wire electrode example to help you choose between the current distribution interfaces in COMSOL Multiphysics® for your electrochemical simulations.
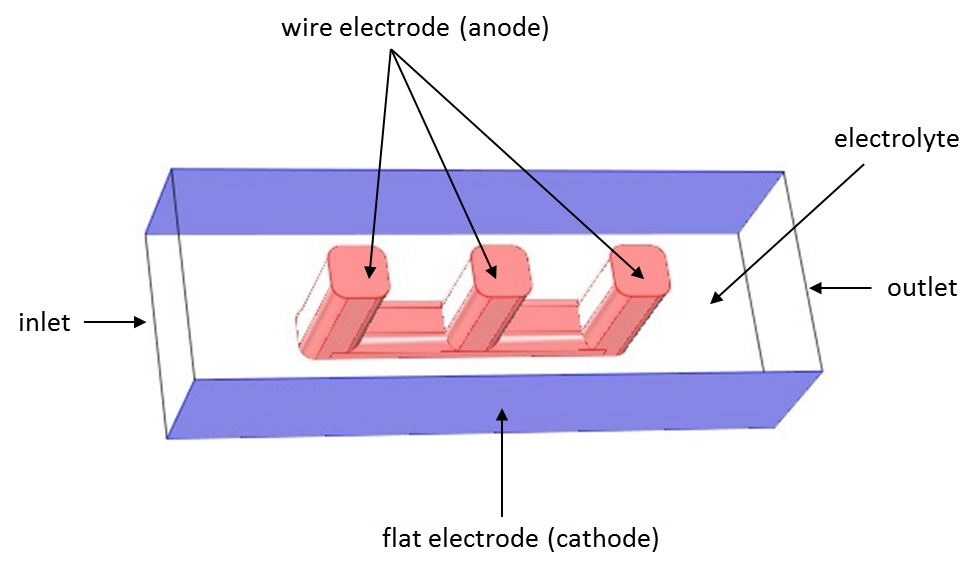
Theory of Current Distribution
Here, we discuss one of the building blocks that make up hybrid parallel computing, namely shared memory computing, as well as when and how to use shared memory with COMSOL Multiphysics®.
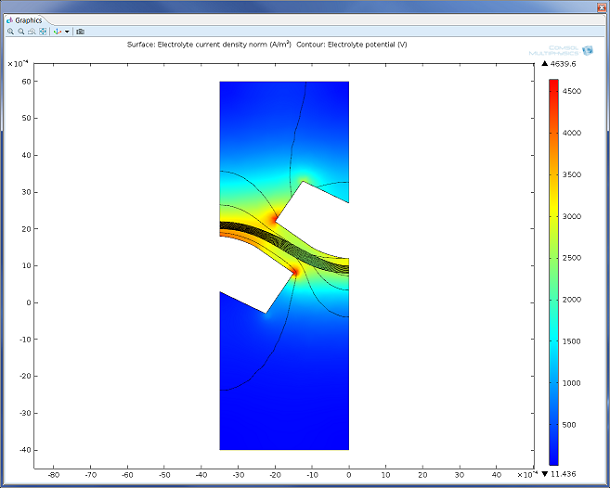
Electrochemistry, from Electroanalysis to Industrial Electrolysis
My colleague, Edmund Dickinson, recently blogged about cyclic voltammetry, and how this can be modeled. It was a fantastic blog entry, as it really described the application, and how to implement such models in COMSOL Multiphysics. While Edmund has a background in electroanalysis, where cyclic voltammetry, potentiometry, and electrochemical impedance are important tools, I had a different but similar life before COMSOL, working within industrial electrolysis. For both of us, the new Electrochemistry Module would have been the perfect tool […]



