Corrosion & Corrosion Protection Blog Posts

The Science Behind Preserving Museum Ships
Managing corrosion and decay are just 2 of the challenges associated with preserving historic vessels. Learn about the approaches that are being used to help keep these ships true to their best-known design.

Modeling Stray Currents from a Light Rail Transit System
The rails in LRT systems can induce stray currents, which may corrode nearby underground metallic structures. Learn how modeling can be used to help.

Modeling Pitting Corrosion in COMSOL Multiphysics®
When the process of generalized corrosion is allowed to continue, the initialization of a pit may occur on the metal surface, which is also referred to as the Evans drop experiment.
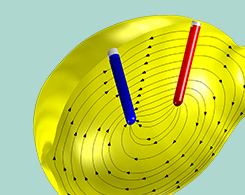
Approaching an Electrochemical Model from Scratch: Lemon Battery
The lemon battery: A high school chemistry experiment, and a great example when learning the general process for how to set up electrochemistry and battery models from scratch.
6 Ways Engineers Are Using Simulation to Help the Environment
Energy-efficient buildings and appliances. Safe nuclear waste storage. Well-preserved freshwater lakes. These are just a few examples of how simulation is being used to help the environment.
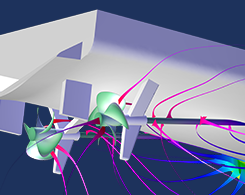
How to Simulate Impressed Current Cathodic Protection
2 common methods for protecting metal structures against galvanic corrosion are sacrificial anode cathodic protection (SACP) and impressed current cathodic protection (ICCP).
Protecting Against Atmospheric Corrosion with Simulation
Certain environmental factors, such as humidity and snow, can lead to atmospheric corrosion. The result? Rusty bikes, cars, and other metal structures. Simulation can protect against this effect.
Multiphysics Modeling of Stress Corrosion in Underground Pipelines
Stress corrosion is a phenomenon that causes degradation in underground pipelines. Learn how to use multiphysics modeling to understand and predict its occurrence.



