COMSOL Events Calendar
Modeling Workflow
Electromagnetics
Structural & Acoustics
Fluid & Heat
Chemical
Interfacing
General
Time zone:
My Time (EDT)
Host Time
08:00 AM - 08:00 PM
Show as:
Search
Showing events starting between: 8 a.m. – 8 p.m.
Upcoming Events
Loading Events

Apr 30
| 9:00 a.m. BST
Modeling Biochemical Sensors and Testing Devices with COMSOL®
COMSOL
Webinar
Online

May 5
| 8:00 a.m. BST
GPU-Beschleunigung in COMSOL®
COMSOL
Webinar
Online
May 5–8
| 11:00 a.m. EDT
Acoustics Modeling in COMSOL Multiphysics®
COMSOL
Training Course
Online

May 5
| 11:00 a.m. EDT
Multiphysics Modeling for Acoustics Applications, Part 2: Acoustic–Thermal Interaction (Thermoacoustics)
COMSOL
Webinar
Online

May 8
| 12:30 p.m. PDT
COMSOL Simulation Summit: Santa Clara
COMSOL
Summit
Santa Clara

May 12
| 8:00 a.m. CEST
Equation-Based Modeling with COMSOL®
COMSOL
Webinar
Online
May 12–15
| 11:00 a.m. EDT
Fuel Cell & Electrolyzers in COMSOL Multiphysics®
COMSOL
Training Course
Online

May 14
| 9:00 a.m. BST
Introduction to Explicit Dynamics in Version 6.4
COMSOL
Webinar
Online

May 14
| 11:00 a.m. EDT
Modeling Granular Flow with COMSOL Multiphysics®
COMSOL
Webinar
Online

May 19
| 8:00 a.m. CEST
Preparing CAD for Analysis with COMSOL®
COMSOL
Webinar
Online

May 19–20
| 9:00 a.m. EDT
CLEO
Vendor
Exhibition
Charlotte

May 19–22
| 11:00 a.m. EDT
Introduction to COMSOL Multiphysics®
COMSOL
Training Course
Online

May 19
| 11:00 a.m. EDT
Synthetic Multiphysics Data for AI Training
COMSOL
Webinar
Online
May 20
| 9:00 a.m. BST
Modeling Water Treatment Processes in COMSOL Multiphysics®
COMSOL
Webinar
Online

May 26
| 8:30 a.m. CEST
Costruire modelli surrogati in COMSOL Multiphysics®
COMSOL
Webinar
Online

May 26
| 11:00 a.m. EDT
Multiphysics Modeling for Acoustics Applications, Part 3: Acoustic-Flow Interaction (Aeroacoustics)
COMSOL
Webinar
Online

May 28
| 11:00 a.m. EDT
COMSOL Day: MedTech
COMSOL
COMSOL Day
Online

Jun 3
| 10:40 a.m. EDT
Battery Modeling with COMSOL Multiphysics®
Tech Briefs
Webinar
Online

Jun 7–9
| 9:00 a.m. EDT
IMS
Vendor
Exhibition
Boston

Jun 8–12
| 11:00 a.m. PDT
AIAA Aviation Forum
COMSOL
Exhibition
San Diego

Jun 9
| 8:00 a.m. CEST
Modeling Electric Motors & Generators
COMSOL
Webinar
Online

Jun 23
| 8:00 a.m. CEST
CFD Modeling with COMSOL® in 18 Minutes
COMSOL
Webinar
Online

Jun 23–26
| 11:00 a.m. EDT
Introduction to COMSOL Multiphysics®
COMSOL
Training Course
Online

Jun 30
| 8:30 a.m. CEST
Modellare il comportamento dei materiali granulari con COMSOL Multiphysics®
COMSOL
Webinar
Online

Jul 7
| 8:00 a.m. CEST
The Basics of COMSOL® in 18 Minutes
COMSOL
Webinar
Online

Jul 14
| 8:00 a.m. CEST
How to Build a Customized Mesh in COMSOL Multiphysics®
COMSOL
Webinar
Online

Jul 21–24
| 11:00 a.m. EDT
Introduction to COMSOL Multiphysics®
COMSOL
Training Course
Online
Aug 4
| 8:00 a.m. CEST
Solving Large Models in COMSOL Multiphysics®
COMSOL
Webinar
Online

Aug 18
| 8:00 a.m. CEST
Thermomechanical Modeling with COMSOL® in 18 Minutes
COMSOL
Webinar
Online

Aug 25–28
| 11:00 a.m. EDT
AC/DC Modeling in COMSOL Multiphysics®
COMSOL
Training Course
Online

Sep 1
| 8:00 a.m. CEST
GPU Acceleration in COMSOL®
COMSOL
Webinar
Online

Sep 1–4
| 11:00 a.m. EDT
CFD Modeling in COMSOL Multiphysics®
COMSOL
Training Course
Online

Sep 15
| 8:00 a.m. CEST
Design Optimization for Engineering Problems
COMSOL
Webinar
Online

Sep 15
| 10:00 a.m. EDT
Introduction to Structural Analysis using COMSOL Multiphysics®
COMSOL
Training Course
Online

Oct 7–9
| 8:00 a.m. EDT
COMSOL Conference 2026 Boston
COMSOL
Conference
Boston
Oct 11–14
| 8:00 a.m. EDT
Society of Engineering Science Technical Meeting 2026
COMSOL
Exhibition
West Lafayette

Oct 20–22
| 7:00 p.m. JST
COMSOL Conference 2026 Tokyo
COMSOL
Conference
Tokyo
Showing 8 of 37 events
Showing 37 of 37 events
| Date | Time | Title | Type | Host | Discipline |
|---|---|---|---|---|---|
| Apr 30 | 9:00 a.m. |
Modeling Biochemical Sensors and Testing Devices with COMSOL® |
Webinar | COMSOL-Online |
|
| May 5 | 8:00 a.m. |
GPU-Beschleunigung in COMSOL® |
Webinar | COMSOL-Online |
|
| May 5–8 | 11:00 a.m. |
Acoustics Modeling in COMSOL Multiphysics® |
Training Course | COMSOL-Online |
|
| May 5 | 11:00 a.m. |
Multiphysics Modeling for Acoustics Applications, Part 2: Acoustic–Thermal Interaction (Thermoacoustics) |
Webinar | COMSOL-Online |
|
| May 8 | 12:30 p.m. |
COMSOL Simulation Summit: Santa Clara |
Special Event | COMSOL-Santa Clara |
|
| May 12 | 8:00 a.m. |
Equation-Based Modeling with COMSOL® |
Webinar | COMSOL-Online |
|
| May 12–15 | 11:00 a.m. |
Fuel Cell & Electrolyzers in COMSOL Multiphysics® |
Training Course | COMSOL-Online |
|
| May 14 | 9:00 a.m. |
Introduction to Explicit Dynamics in Version 6.4 |
Webinar | COMSOL-Online |
|
| May 14 | 11:00 a.m. |
Modeling Granular Flow with COMSOL Multiphysics® |
Webinar | COMSOL-Online |
|
| May 19 | 8:00 a.m. |
Preparing CAD for Analysis with COMSOL® |
Webinar | COMSOL-Online |
|
| May 19–20 | 9:00 a.m. |
CLEO |
Special Event | COMSOL-Charlotte |
|
| May 19–22 | 11:00 a.m. |
Introduction to COMSOL Multiphysics® |
Training Course | COMSOL-Online |
|
| May 19 | 11:00 a.m. |
Synthetic Multiphysics Data for AI Training |
Webinar | COMSOL-Online |
|
| May 20 | 9:00 a.m. |
Modeling Water Treatment Processes in COMSOL Multiphysics® |
Webinar | COMSOL-Online |
|
| May 26 | 8:30 a.m. |
Costruire modelli surrogati in COMSOL Multiphysics® |
Webinar | COMSOL-Online |
|
| May 26 | 11:00 a.m. |
Multiphysics Modeling for Acoustics Applications, Part 3: Acoustic-Flow Interaction (Aeroacoustics) |
Webinar | COMSOL-Online |
|
| May 28 | 11:00 a.m. |
COMSOL Day: MedTech |
COMSOL Day | COMSOL-Online |
|
| Jun 3 | 10:40 a.m. |
Battery Modeling with COMSOL Multiphysics® |
Webinar | Tech Briefs - Online |
|
| Jun 7–9 | 9:00 a.m. |
IMS |
Special Event | COMSOL-Boston |
|
| Jun 8–12 | 11:00 a.m. |
AIAA Aviation Forum |
Special Event | COMSOL-San Diego |
|
| Jun 9 | 8:00 a.m. |
Modeling Electric Motors & Generators |
Webinar | COMSOL-Online |
|
| Jun 23 | 8:00 a.m. |
CFD Modeling with COMSOL® in 18 Minutes |
Webinar | COMSOL-Online |
|
| Jun 23–26 | 11:00 a.m. |
Introduction to COMSOL Multiphysics® |
Training Course | COMSOL-Online |
|
| Jun 30 | 8:30 a.m. |
Modellare il comportamento dei materiali granulari con COMSOL Multiphysics® |
Webinar | COMSOL-Online |
|
| Jul 7 | 8:00 a.m. |
The Basics of COMSOL® in 18 Minutes |
Webinar | COMSOL-Online |
|
| Jul 14 | 8:00 a.m. |
How to Build a Customized Mesh in COMSOL Multiphysics® |
Webinar | COMSOL-Online |
|
| Jul 21–24 | 11:00 a.m. |
Introduction to COMSOL Multiphysics® |
Training Course | COMSOL-Online |
|
| Aug 4 | 8:00 a.m. |
Solving Large Models in COMSOL Multiphysics® |
Webinar | COMSOL-Online |
|
| Aug 18 | 8:00 a.m. |
Thermomechanical Modeling with COMSOL® in 18 Minutes |
Webinar | COMSOL-Online |
|
| Aug 25–28 | 11:00 a.m. |
AC/DC Modeling in COMSOL Multiphysics® |
Training Course | COMSOL-Online |
|
| Sep 1 | 8:00 a.m. |
GPU Acceleration in COMSOL® |
Webinar | COMSOL-Online |
|
| Sep 1–4 | 11:00 a.m. |
CFD Modeling in COMSOL Multiphysics® |
Training Course | COMSOL-Online |
|
| Sep 15 | 8:00 a.m. |
Design Optimization for Engineering Problems |
Webinar | COMSOL-Online |
|
| Sep 15 | 10:00 a.m. |
Introduction to Structural Analysis using COMSOL Multiphysics® |
Training Course | COMSOL-Online |
|
| Oct 7–9 | 8:00 a.m. |
COMSOL Conference 2026 Boston |
Special Event | COMSOL-Boston |
|
| Oct 11–14 | 8:00 a.m. |
Society of Engineering Science Technical Meeting 2026 |
Special Event | COMSOL-West Lafayette |
|
| Oct 20–22 | 7:00 p.m. |
COMSOL Conference 2026 Tokyo |
Special Event | COMSOL-Tokyo |
|
Trainings by Certified Consultants
On-Demand Webinars

Modeling Microwave-Heated Multiphase Reactors Using COMSOL Multiphysics®
Webinar
Apr 28

Modeling Optical Nanostructures with COMSOL Multiphysics®
Webinar
Apr 23

Modellazione multifisica dei dispositivi di comunicazione ad alta velocità
Webinar
Apr 23

Bonnes pratiques pour les études et les solveurs avec COMSOL Multiphysics®
Webinar
Apr 23

COMSOL® 多物理场仿真在半导体键合工艺中的应用
Webinar
Apr 23

Modeling Heat Transfer of Spacecraft with COMSOL Multiphysics®
Webinar
Apr 21
Design and Optimization of Semiconductor Fabrication Equipment Using COMSOL®
Webinar
Apr 16

Modéliser les systèmes de puissance pour le réseau électrique avec COMSOL Multiphysics®
Webinar
Apr 16

借助 GPU 加速 COMSOL Multiphysics® 仿真
Webinar
Apr 16

Improving Thermal Management of Electronic Devices with COMSOL Multiphysics®
Webinar
Apr 15

Multiphysics Modeling for Acoustics Applications, Part 1: Acoustic–Structure Interaction (Vibroacoustics)
Webinar
Apr 14

COMSOL® 中的 CFD 仿真
Webinar
Apr 09

使用 COMSOL® 模拟放电现象
Webinar
Apr 02
使用 COMSOL® 进行形状与拓扑优化
Webinar
Mar 26

Solving Large Models in COMSOL Multiphysics®
Webinar
Mar 25

Multiphysics Simulation for Automotive Power Electronics
Webinar
Mar 25

Analisi acustiche nel settore automobilistico con COMSOL Multiphysics®
Webinar
Mar 24
Comment construire un maillage personnalisé dans COMSOL Multiphysics®
Webinar
Mar 19

COMSOL® 多物理场仿真优化光学超材料设计
Webinar
Mar 19

Innovating Hydrogen Technologies with Simulation
Webinar
Mar 18

Modeling Gyroscopes and Accelerometers in COMSOL Multiphysics®
Webinar
Mar 17

Améliorer les Performances des Piles à Combustible et des Electrolyseurs avec COMSOL Multiphysics®
Webinar
Mar 12

使用 COMSOL® 模拟高性能电机
Webinar
Mar 12
Predicting Viscous Fingering in Porous Media Using COMSOL Multiphysics®
Webinar
Mar 05

使用模型管理器进行仿真数据管理
Webinar
Mar 05

Why Model and Simulate Batteries?
Webinar
Mar 04

Editing, Repairing, and Combining Imported STL Files with CAD
Webinar
Mar 03

Modeling Rotordynamics with COMSOL Multiphysics®
Webinar
Feb 26

The Basics of COMSOL® in 18 Minutes
Webinar
Feb 26
Modélisation géométrique avec COMSOL Multiphysics®
Webinar
Feb 19

Analisi e visualizzazione dei risultati in COMSOL Multiphysics®
Webinar
Feb 18

Modeling Nonlinear Photonics in Dielectric Resonant Metasurfaces Using COMSOL®
Webinar
Feb 18

Modéliser la Corrosion et les Systèmes de Protection contre la Corrosion avec COMSOL Multiphysics®
Webinar
Feb 12
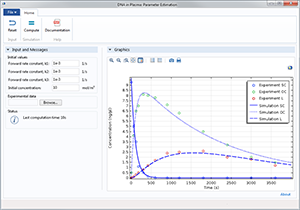
非线性结构材料参数估计
Webinar
Feb 05

Modeling and Simulation of MEMS Devices
Webinar
Feb 04

Modeling Photonic Crystals in COMSOL Multiphysics
Webinar
Jan 29

Modéliser les batteries avec COMSOL Multiphysics®
Webinar
Jan 29

COMSOL® 多物理场仿真在功率半导体中的应用
Webinar
Jan 29

Modeling Emerging Battery Technologies with COMSOL Multiphysics®
Webinar
Jan 28

Modeling Emerging Battery Technologies with COMSOL Multiphysics®
Webinar
Jan 28

Modéliser les Bobines avec COMSOL Multiphysics®
Webinar
Jan 22
Automotive Acoustics Analysis Using COMSOL Multiphysics
Webinar
Jan 21

Modellare i flussi turbolenti con COMSOL Multiphysics®
Webinar
Jan 20

COMSOL® 中的流热耦合仿真
Webinar
Jan 15

在 COMSOL® 中实现仿真流程自动化
Webinar
Jan 08

多物理场仿真结果的处理与可视化
Webinar
Dec 25

COMSOL Multiphysics® 6.4 版本功能简介
Webinar
Dec 18

Parameter Estimation for Nonlinear Materials
Webinar
Dec 11
Modéliser le contact mécanique avec COMSOL Multiphysics®
Webinar
Dec 11

COMSOL® 中的等离子体仿真
Webinar
Dec 11

Introducing COMSOL Multiphysics® Version 6.4
Webinar
Dec 10

Migliorare la progettazione e la gestione termica delle batterie
Webinar
Dec 10

Modéliser les dispositifs médicaux et biomédicaux avec COMSOL Multiphysics®
Webinar
Dec 04

COMSOL® 中的化学反应工程仿真
Webinar
Dec 04

Multiphysics Simulation for Vehicle Electrification
Webinar
Dec 02

COMSOL® 多物理场仿真在新能源汽车领域中的应用
Webinar
Nov 27

Optimising Manufacturing Processes Through Simulation
Webinar
Nov 26

Simulare per il biomedicale con COMSOL Multiphysics®
Webinar
Nov 26

Designing Efficient Electric Motors with COMSOL Multiphysics
Webinar
Nov 26

使用 COMSOL® 模拟声学超材料
Webinar
Nov 20

Efficient Battery Modeling with Surrogate Models
Webinar
Nov 19

Automating Your Modeling Workflow in COMSOL Multiphysics®
Webinar
Nov 18

Modéliser le packaging et les tests de dispositifs électroniques avec COMSOL Multiphysics®
Webinar
Nov 13

使用 COMSOL 模拟 RF 和光学周期性结构
Webinar
Nov 13

Progettare per l’industria aerospaziale con COMSOL Multiphysics®
Webinar
Nov 05

Modeling Gyroscopes and Accelerometers in COMSOL Multiphysics®
Webinar
Oct 30

COMSOL 多物理场仿真在功率器件中的应用
Webinar
Oct 23

Personalizza le tue equazioni con COMSOL Multiphysics®
Webinar
Oct 22

Modéliser les écoulements turbulents avec COMSOL Multiphysics®
Webinar
Oct 16

COMSOL 多物理场仿真在数字岩心中的应用
Webinar
Oct 16

Modeling and Simulation for Electric Motor Design
Webinar
Oct 15

Simulating Electrical Power Systems for the Grid
Webinar
Oct 14

Unravelling Chemomechanical Effects in Lithium-Ion Batteries
Webinar
Oct 09
Optimierung keramischer 3D-Drucke durch Simulation
Webinar
Oct 09

Simulare sistemi elettrici di potenza
Webinar
Oct 08

Modeling Next-Generation Battery Technologies with COMSOL Multiphysics®
Webinar
Oct 06

Modeling Emerging Battery Technologies with COMSOL Multiphysics®
Webinar
Sep 25

Modéliser le rayonnement thermique avec COMSOL Multiphysics®
Webinar
Sep 25
在 COMSOL 中创建、训练和使用代理模型
Webinar
Sep 25

Ottimizzare le simulazioni di meccanica strutturale
Webinar
Sep 24

Modeling and Simulation of MEMS Devices
Webinar
Sep 23

Elektrische Entladungen modellieren
Webinar
Sep 18

Modéliser les moteurs électriques et les générateurs avec COMSOL Multiphysics®
Webinar
Sep 18

电力设备噪声的多物理场仿真
Webinar
Sep 18
Costruire app di simulazione e digital twin con COMSOL Multiphysics®
Webinar
Sep 16

Modeling Reaction Kinetics and Pharmacokinetics with COMSOL Multiphysics®
Webinar
Sep 11

Optimization of Thermal Management Systems
Webinar
Sep 03

Prediction of Electromagnetic Signatures of Naval Vessels with COMSOL Multiphysics
Webinar
Aug 28

Modellare sistemi ottici con COMSOL Multiphysics®
Webinar
Aug 28

Bonnes pratiques pour évaluer et afficher les résultats dans COMSOL Multiphysics®
Webinar
Aug 28

COMSOL 多物理场仿真在声学中的应用
Webinar
Aug 27

COMSOL 仿真 App 开发、部署和应用
Webinar
Aug 21

Modeling and Simulation for Electric Motor Design
Webinar
Aug 14

使用 COMSOL 模拟热应力
Webinar
Aug 14

COMSOL 在半导体封测中的应用
Webinar
Aug 07

Modeling Hydrogen Fuel Cells and Electrolyzers
Webinar
Aug 06

Modeling Acoustic Metamaterials in COMSOL Multiphysics
Webinar
Jul 31

使用 COMSOL 模拟多孔介质流和地下水流
Webinar
Jul 31

Fatigue and Durability Analysis Using COMSOL Multiphysics®
Webinar
Jul 24

Comment créer des modèles de substitution avec COMSOL Multiphysics®
Webinar
Jul 24

多物理场仿真在电力变压器中的应用
Webinar
Jul 24

Biomedical Modeling with COMSOL Multiphysics
Webinar
Jul 17
COMSOL 多物理场仿真在光学领域中的应用
Webinar
Jul 16
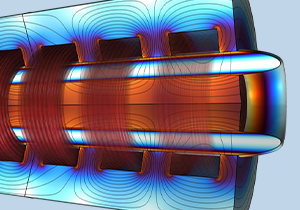
Understanding Liquid Metal Transport in Magnetic Fields — Simulating Magnetohydrodynamic Duct Flow
Webinar
Jul 11

Modeling Electric Motors and Drivetrains
Webinar
Jul 10

Créer des applications de simulation et des jumeaux numériques avec COMSOL Multiphysics®
Webinar
Jul 10

多物理场仿真在半导体制程中的应用
Webinar
Jul 10

COMSOL® 中的低频电磁场仿真
Webinar
Jul 03

Memristor Modeling with COMSOL Multiphysics®
Webinar
Jul 02

使用 COMSOL 进行流固耦合仿真
Webinar
Jun 26

COMSOL® 中的传热仿真
Webinar
Jun 19

Modellierung optischer Nanostrukturen mit COMSOL Multiphysics®
Webinar
Jun 17

Optimization in Structural Mechanics
Webinar
Jun 12

Hydrogen Technology Innovation with Simulation
Webinar
Jun 12

Modeling Hydrogen Fuel Cells and Electrolyzers
Webinar
Jun 12

Améliorer la gestion thermique des dispositifs électroniques avec COMSOL Multiphysics®
Webinar
Jun 12

多物理场仿真在先进封装中的应用
Webinar
Jun 12

Modeling Chemical Reactions and Reacting Flows Using COMSOL Multiphysics®
Webinar
Jun 05

COMSOL® 在电力设备中的应用
Webinar
Jun 05

Migliorare le tecnologie per la decarbonizzazione con la simulazione
Webinar
Jun 04

COMSOL® 网格划分功能介绍
Webinar
May 29

Introduction to Multibody Dynamics Modeling with COMSOL Multiphysics
Webinar
May 28

Automotive Acoustics Analysis Using COMSOL Multiphysics®
Webinar
May 27

Comment faire de l’optimisation et de l’estimation de paramètres avec COMSOL Multiphysics®
Webinar
May 22

COMSOL® 在半导体薄膜沉积工艺中的应用
Webinar
May 20
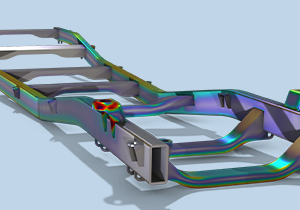
COMSOL® 中的结构力学仿真
Webinar
May 15
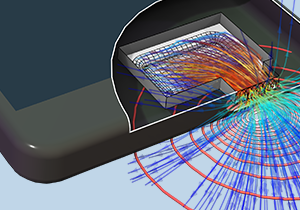
Analizzare i fenomeni acustici con la simulazione
Webinar
May 14

Modélisation multiphysique pour l’aéronautique et le spatial avec COMSOL Multiphysics®
Webinar
May 13

Improving Thermal Management of Batteries with COMSOL Multiphysics®
Webinar
May 09

Demokratisierung der Multiphysik-Simulation
Webinar
May 08

COMSOL® 中的 CFD 仿真
Webinar
May 08

将随频率变化的数据融入 COMSOL® 时域声学仿真
Webinar
Apr 24

COMSOL® 中的电池仿真
Webinar
Apr 17

使用 COMSOL® 模拟生物传感器和检测设备
Webinar
Mar 27
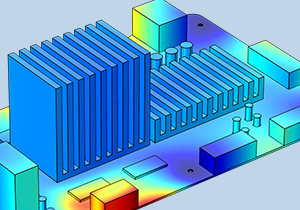
Multiphysics Modeling of Electronic Components
Webinar
Mar 20

使用 COMSOL® 模拟电声器件
Webinar
Mar 20

使用 COMSOL® 进行锂离子电池模型的参数估计
Webinar
Mar 06

使用 COMSOL® 模拟放电现象
Webinar
Feb 27
使用 COMSOL® 模拟雷达和天线
Webinar
Feb 20

使用模型管理器进行仿真数据管理
Webinar
Feb 13

LiveLink™ for MATLAB® 和 LiveLink™ for Simulink® 功能简介
Webinar
Jan 09

COMSOL® 中的低频电磁场仿真
Webinar
Jan 03

使用 COMSOL Multiphysics® 优化设计
Webinar
Dec 26

COMSOL® 中的结构力学仿真
Webinar
Dec 19

COMSOL® 中的传热仿真
Webinar
Dec 12

COMSOL® 中的多相流仿真
Webinar
Dec 05

COMSOL Multiphysics® 6.3 版本功能简介
Webinar
Nov 28

COMSOL® 中的转子动力学和多体动力学仿真
Webinar
Nov 21

COMSOL® 求解器介绍及设置方法
Webinar
Nov 14
使用 COMSOL Multiphysics® 模拟声学超材料
Webinar
Nov 07

多物理场仿真结果的处理与可视化
Webinar
Oct 24

COMSOL® 中的等离子体仿真
Webinar
Oct 17

COMSOL® 网格划分功能介绍
Webinar
Oct 10

电力变压器的多物理场仿真
Webinar
Sep 19

使用 COMSOL Multiphysics® 仿真高性能电机
Webinar
Sep 12

不确定性量化在多物理场仿真中的应用
Webinar
Sep 05

超声波及其应用的多物理场仿真
Webinar
Aug 29
使用 COMSOL Multiphysics® 模拟光电半导体
Webinar
Aug 15

COMSOL® 多物理场仿真加速数字岩心技术发展
Webinar
Aug 08

使用代理模型开发高效的 COMSOL® 仿真 App
Webinar
Aug 01

COMSOL® 多物理场仿真在地热能开发中的应用
Webinar
Jul 25

COMSOL® 仿真 App 开发、部署和应用
Webinar
Jul 18

COMSOL® 中的光学仿真
Webinar
Jul 11
非线性结构材料参数估计
Webinar
Jul 04

基于 COMSOL® 构建电力设备的数字孪生
Webinar
Jun 27
COMSOL® 室内声学仿真
Webinar
Jun 20

COMSOL Multiphysics® 在半导体制造中的应用
Webinar
Jun 13

COMSOL Multiphysics® 在绿氢技术中的应用
Webinar
Jun 06

使用 COMSOL Multiphysics® 模拟放电现象
Webinar
May 28
使用 COMSOL® 模拟 SAW/BAW 器件
Webinar
May 16
COMSOL® 中的换热器仿真
Webinar
May 09
Showing 6 of 171 events
Showing 171 of 171 events
Trainings by Certified Consultants
- Product Information
- Products
- Specification Chart
- License Options
- System Requirements
- Release History
- Support and Services
- Support Center
- My Support Cases
- Knowledge Base
- Partners and Consultants
- Documentation
- Product Download
- Company
- About
- Careers
- Press
- Contact Us
- |
- Privacy Policy
- |
- Trademarks
- |
- Cookie Settings
- © 2026 by COMSOL. All rights reserved



