
Electrodeposition is the process of making a substance adhere to an object through electrochemical reactions. Sometimes the substance is available in the solution form and other times it is a solid object too, and needs to undergo electrochemical reactions in order to dissolve into solution; often as part of the electrodeposition process. Electrodeposition can be an important part of the refining process of certain metals, such as copper, silver, and gold and is often referred to as electrorefining or electrowinning. Another form of electrodeposition is known as “electroplating”.
What is Electroplating?
You may have wondered why car rims and motorcycle parts are so shiny, and the answer is: electroplating. Generally speaking, electroplating is used for many purposes, including augmenting undersized parts and altering an object’s surface properties. During the process, metal often goes to waste and companies need to figure out how to reduce the amount of metals that are consumed. Many times, this waste occurs as electrodeposited layers are applied unevenly, and an excess of material is required to ensure complete coverage. PEM is one such company, and by using COMSOL they figured out how to reduce the metal deposit during electrolysis by 10-30%. Several parameters are important when engaging in electroplating, and COMSOL’s Electrodeposition Module can be used to study these to ensure the desired outcome of the electrochemical process.
Modeling Decorative Plating
We have two tutorial models for the Electrodeposition Module that touch on electroplating. One of them is a decorative plating model, demonstrating how to use COMSOL to calculate the thickness of a deposited layer as well as the pattern created by the dissolution of the anode surface. The electroplating model is a secondary current density distribution model and uses full Butler-Volmer kinetics for the anode and the cathode. The planar anode dissolves, while the cathode is a furniture fitting of sorts that is to be decorated with a nice finishing metal. You can study the following important parameters using the Electrodeposition Module:
- Cell geometry
- Electrolyte composition and mixing
- Electrode kinetics
- Operating potential and average current density
- Temperature
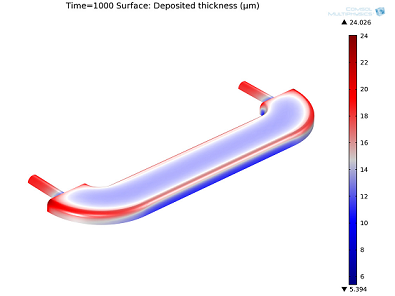
Electroplating: Thickness of the decorative layer in a furniture fitting.
Simulations based on secondary current distribution.
A Tricky Business Made Simple with Multiphysics Simulation
The company based out of France, PEM, took this type of modeling to the next level, when they also considered the effects of fluid flow and convection. They are in the business of treating and finishing surfaces for the electronics industry and make this a viable process through reel-to-reel electroplating. The customer provides PEM with the reel containing tape holding a series of metal parts and PEM then unwinds the tape from the reel and sends it through an electroplating process. Here, the tape is turned into the cathode via contact with a voltage source, and the anodes are located in the plating cell. The electrical circuit is closed by an electrolyte. The big trick is to achieve a uniform plating layer on the tape as edge effects cause more metal to accumulate around the edges of the tape. PEM’s solution to this problem was to design a shield of sorts to even out the plating across the width of the tape.
The shape of this shield is dependent on the reel’s components, the plating material, the thickness of the plating material, the concentration of the electrolyte, and the traveling speed of the tape. Each new job or batch-run can first be simulated to design an appropriate shield, through COMSOL Multiphysics. They found that computer modeling eliminated the need for time-consuming experiments and physical prototypes — with a successful design now often reached on the first try! Next, PEM used COMSOL to design more efficient reactor cells. As Dr. Philippe Gendre of PEM states in COMSOL News: “If you can replenish the metal ions better, you can raise the current density and thus production.”
This was back in 2008, and these days we have the Electrodeposition Module to make these types of calculations even easier to perform.






Comments (0)