
Currently, the most common methods for treating cancer — surgery, chemotherapy, and radiation therapy — carry significant risks. A potentially safer alternative is electrochemical treatment (EChT), which uses direct current to eliminate malignant tumors. Before this technique can be widely used, though, doctors need a better understanding of the destruction mechanics so that they can develop dose-planning strategies.
The Advantages and Challenges of Electrochemical Tumor Treatment
Compared to other treatment methods, EChT offers several potential advantages:
- Safety
- Effectiveness
- Local effect
- Minimal invasiveness
Further, this technique can treat many tumors (both benign and malignant) that traditional methods cannot, such as tumors that are inoperable by surgery as well as those that aren’t responsive to chemo or radiation.

EChT shows promise as a way to remove tumors in cancer patients. Image by Dr med Peter Wolf from Innovations in biological cancer therapy, a guide for patients and their relatives, Naturasanitas 2008, ISBN 978-3-9812416-1-7. Licensed under CC BY-SA 3.0, via Wikimedia Commons.
Another benefit of EChT is that it’s a fairly simple process. In short, two or more electrodes are inserted into (or near) the tumorous tissue and a direct current is applied. The current causes oxygen and chlorine evolution at the anode as well as hydrogen evolution at the cathode. These electrochemical reactions in the tumor make the tissue, which contains salty water, much more acidic and alkaline near the anode and cathode, respectively. The tumor cells are then rapidly destroyed by the new, extreme pH values.
What’s stopping EChT from becoming a common treatment for tumors? One major obstacle is the uncertainty surrounding this method, in particular exactly how the tissue is destroyed. For instance, scientists know that pH levels can be lowered by oxygen evolution (which creates positive hydrogen ions) and chlorine production (via hydrolysis) at the anode. However, the role each of these reactions plays and how they contribute to tumor eradication isn’t clear.
Without a better understanding of tumor destruction mechanisms, it is challenging to develop effective dose-planning strategies for tumor treatment. To gain the insight needed, one option is investigating EChT using the COMSOL Multiphysics® software and one of the following add-on products:
Let’s look at a simple example that evaluates the electrochemical reactions occurring near the anode during EChT.
Modeling EChT with COMSOL Multiphysics®
This example consists of an electrode inside tumor tissue in the liver. The model is simplified to 1D axisymmetry, thanks to the rotational symmetry, and focuses only on the transport and reactions occurring near the anode (oxygen evolution and chlorine production). For this reason, there are only three species that are considered:
- Hydrogen
- Chlorine
- Sodium
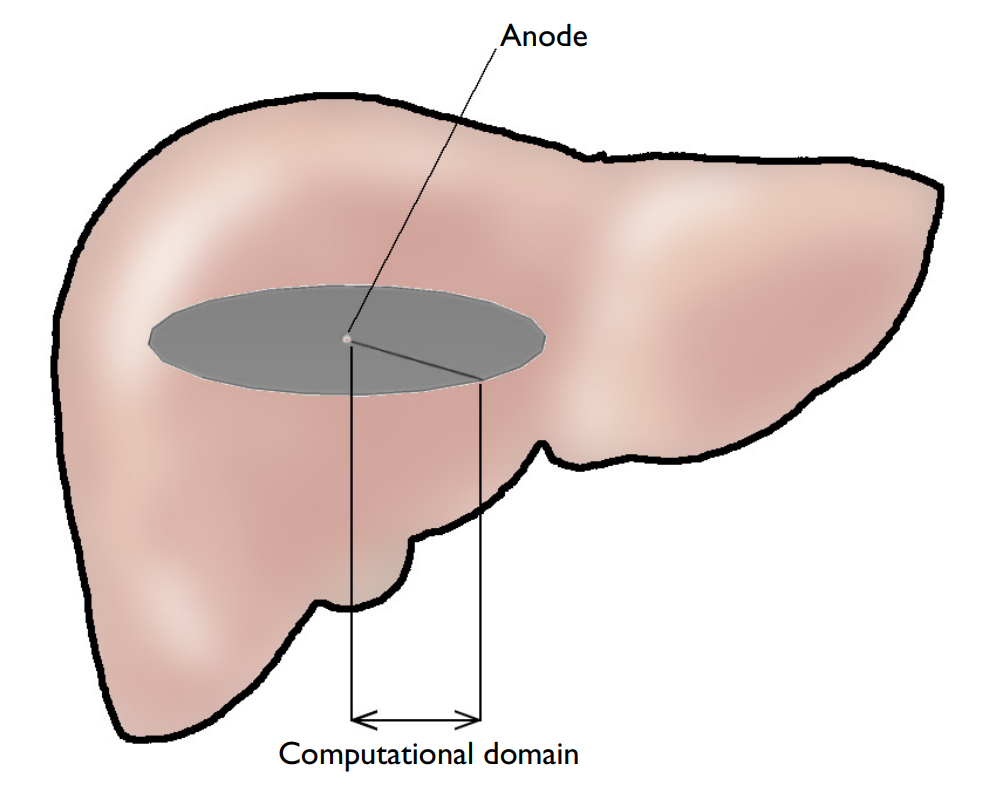
Schematic of the liver, tumor tissue, and anode.
To model the transport of these species, you first need to add the Tertiary Current Distribution, Nernst-Planck interface. This physics interface enables you to model the movement of all of the species through the tumor, which is assumed to take place radially to and from the anode. It can also account for important aspects of the species transport, including the different diffusivities, concentrations, and mobilities (for details about the equations involved, see the Electrochemical Treatment of Tumors tutorial).
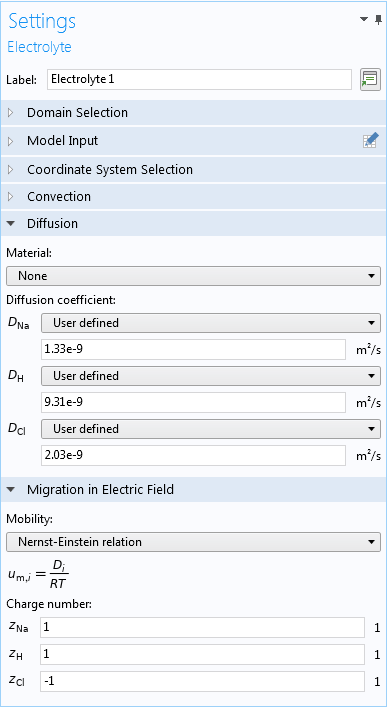
For the electrolyte, you can enter diffusion coefficients manually, or from a material, and specify the mobility from the Nernst-Einstein relation.
Then, you can use the built-in Electrode Surface feature to describe the chlorine and oxygen evolution at the anode’s surface as well as the resulting fluxes. (The sodium does not take part in the electrode reactions.)
Another important aspect of this model is the current density, since it affects the transport of the species and is also affected by the concentrations nearby. For instance, high current densities produce a lot of hydrogen ions close to the electrode. However, the current density doesn’t remain the same throughout EChT, so the production isn’t constant, either. Instead, the current density slowly drops off and protons diffuse away from the electrode surface to areas with a lower concentration.
To describe the varying current density for the reactions, you can add the Electrode Reaction feature and then include concentration-dependent kinetic expressions based on parameters such as the exchange current density, anodic and cathodic transfer coefficients, as well as reduced and oxidized species expressions.
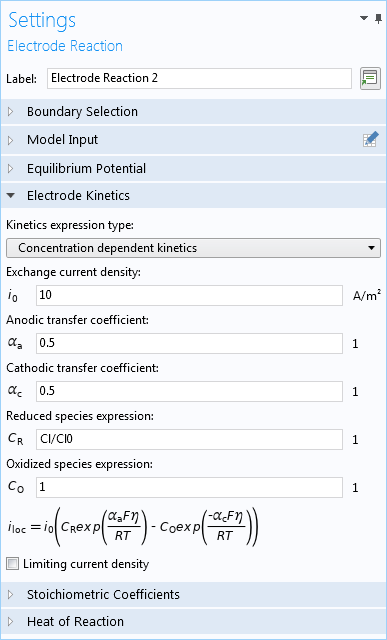
The electrode reaction is defined for chlorine using concentration-dependent kinetic expressions.
Gaining Insight into the Electrolysis of Tumor Tissue
First up in the results, you can see how long it takes for the reactions to lower the pH level enough to start to destroy the tumor (2 or below, according to experimental and theoretical findings). For this example, the level becomes appropriately acidic around 3000 seconds.
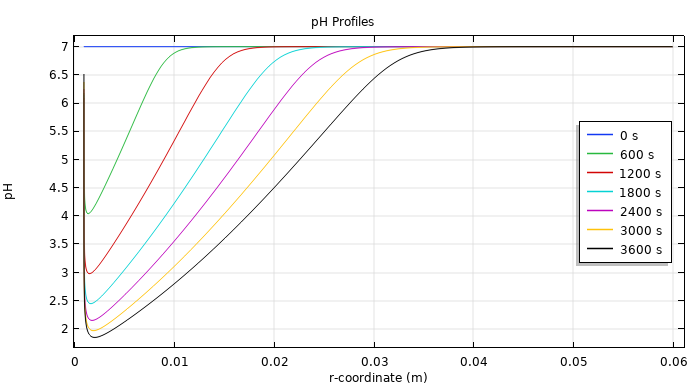
Plot of the pH levels at different time steps during EChT.
You can also examine the concentration levels of hydrogen ions (top image below) and chloride (bottom image below) near the anode, as this provides insight into their transport. The maximum concentration of hydrogen ions occurs a short distance away from the anode, which is due to the electroneutrality condition, since the chloride ions are depleted close to the anode. As for the chloride, the concentration close to the anode steadily decreases. This leads to a drop in chloride production and, in turn, a higher oxygen evolution rate.
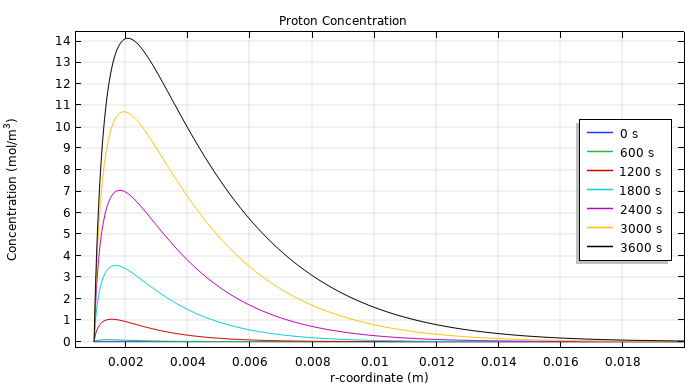
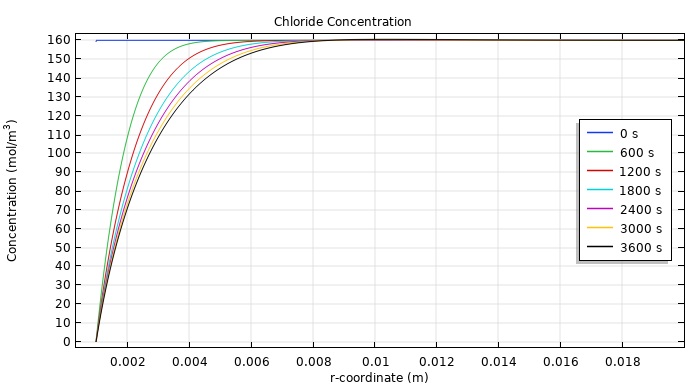
The concentrations of hydrogen (top) and chloride (bottom) in the tissue at different time steps. Note that these images only show the concentration up to 0.02 meters away from the anode.
You can evaluate how the total current density, oxygen evolution, and chlorine production relate to each other by plotting them together, as shown below. Here, as the chloride concentration at the electrode surface drops, the concentration overvoltage for its production increases and causes a sharp dip in the total current density. As mentioned previously, though, the chloride production soon decreases while the oxygen evolution increases, leading to a slow but steady rise in the electrode current density.
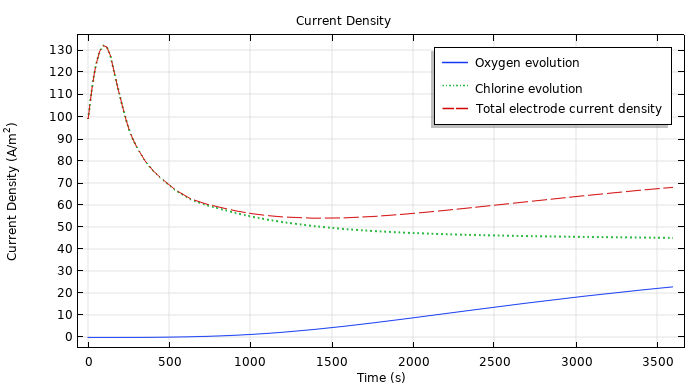
The total current density, chlorine production, and oxygen evolution during EChT.
As demonstrated by this simple example, biomedical scientists can use mathematical modeling to learn more about the mechanics of EChT, which could help them develop dose-planning methods. An extension of this model could include the reactions at the cathode or investigate the effects of different voltages.
Next Steps
Try the EChT example discussed here by clicking the button below. Then, you can download the PDF documentation for the model and the MPH file.
Learn more about how simulation can advance cancer treatment in these blog posts:






Comments (0)